Elevating Compliance and GxP in regulated industries
ComplianceWire helps scale trainings to meet regulatory and industry standards in a FDA CFR Part 11 validated system. Navigate regulations, automate training and implement risk management to support development of safer and more effective medical products.
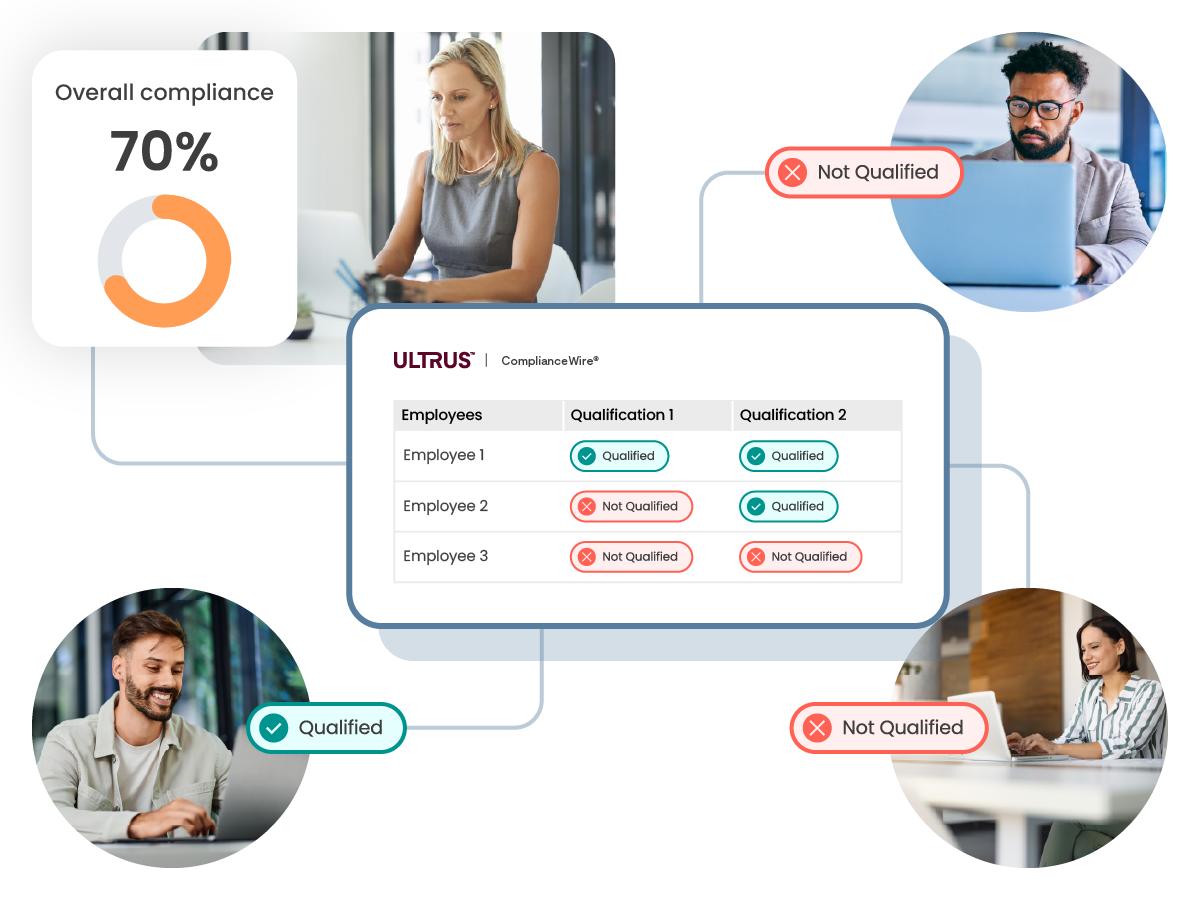
Gain oversight into your employee qualifications and training
Track and monitor the status of your employees in meeting their required qualifications and training in aggregate, or drill into the areas where improvement possibilities and risks are identified.
Qualification overview
Dashboards and reports provide an organization-wide view of compliance and qualification status.
Qualification matrix
Drill down into each team and qualification in real time to understand exactly what is missing that is preventing compliance.
Schedule reports
Schedule reports to team leaders to support the active promotion of training and qualifications.
Streamline and automate your employee qualification process
Easily communicate with and monitor your employees and follow up on post-audit findings (CAPAs) and other regulatory obligations. Upload required training, qualifications and policies to maintain compliance.
Create and augment
Easily create your own courses or edit and re-brand our off-the-shelf library to cater the content to your organization.
Automate training
Users can easily engage in dynamic eLearning courses to scale up training.
Fully configurable
Create different mechanisms for standards to meet compliance, build out team hierarchies and integrate with your wider ecosystem.
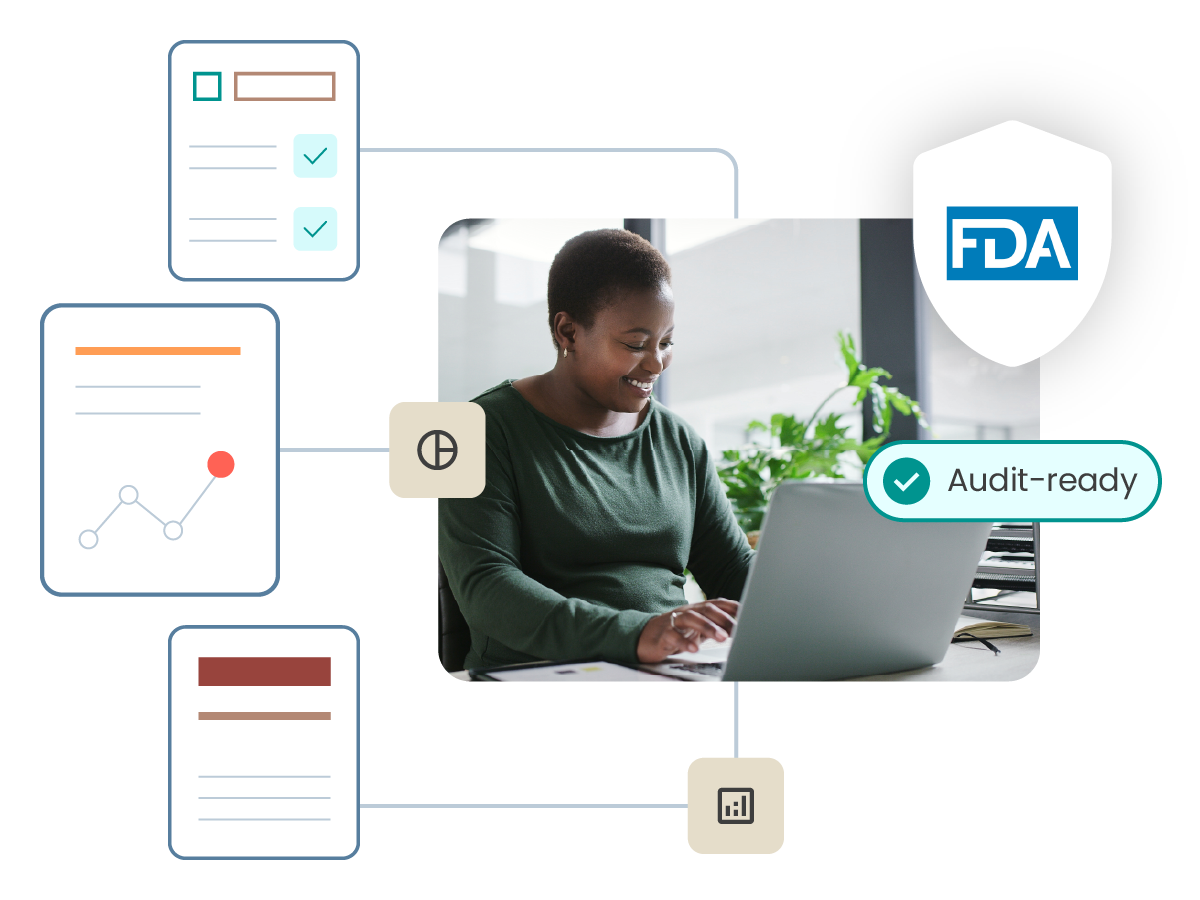
Be ready to respond to an audit or regulatory inquiry
ComplianceWire is natively compliant with FDA 21 CFR Part 11 standards and contains many of the requested reports from regulatory agencies right out of the box. Respond to authorities and internal audits confidently.
Validated system
Learn how UL Solutions verifies every major release of ComplianceWire®, with insights from our Strategic Advisory experts.
Audit readiness support
Leverage the ComplianceWire team to prepare for and support regulatory inquiries.
Proven trust
ComplianceWire is regularly audited internally and by our customers. On average, our customers audit ComplianceWire over 50 times a year, including paper, remote and onsite audits.
Grow expertise with ComplianceWire’s e-Learning Library for Life Sciences
Get help to remain compliant with the U.S. Food and Drug Administration (FDA) regulation of medical devices and pharmaceuticals. Our course catalog contains more than 400 life sciences e-learning courses, many of which were co-developed with the FDA.
Extensive coverage
UL Solutions offers over 1,000 standard regulatory and knowledge-focused e-learning courses, including 21 CFR part 820 training, 508 compliance e-learning, CAPA training and more.
Regulatory expertise
UL Solutions expertise combined with FDA-authored and/or reviewed courses, identical to those used by the FDA to train its inspectors and investigators.
Engaging content
Includes stylized topic-specific videos, embedded interactions, and quizzes, all implemented by skilled instructional designers.
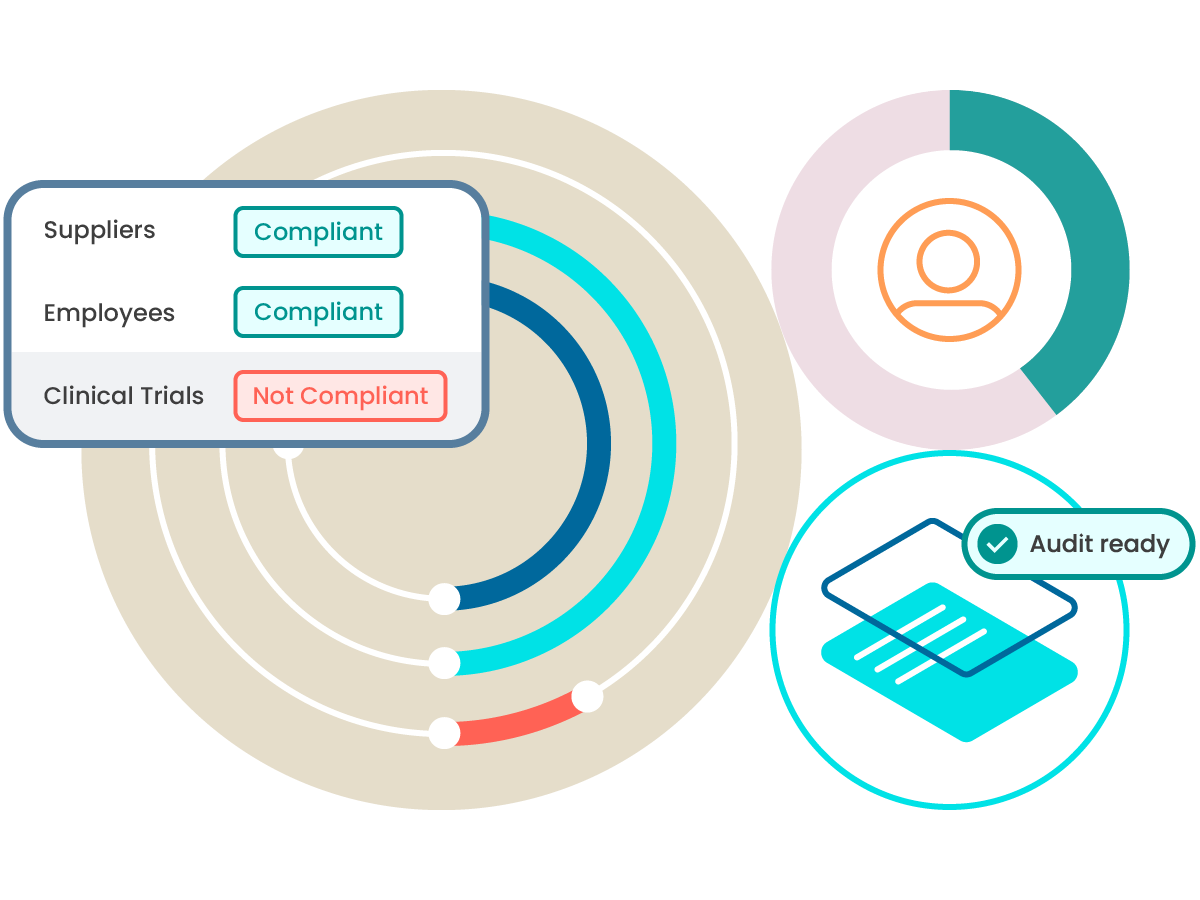
Scale quality and compliance across your business processes
Elevate quality from being a manufacturing-only focus to being a central pillar that helps reduce risk across the entire business.
Supplier qualification
Learn how ComplianceWire can help support qualification and quality across your supply chain.
Clinical trials
Support positive patient outcomes and help clinical research by covering qualifications across clinical trials.
Social responsibility
Help support employees, suppliers, partners and vendors who are meeting the standards of excellence associated with your brand.
ComplianceWire® and ULTRUS™ software
ComplianceWire® is now part of ULTRUS software, which brings together flagship digital offerings from UL Solutions to help customers manage their regulatory, supply chain and sustainability challenges.
Frequently Asked Questions
- What types of regulated industries typically use ComplianceWire®?
ComplianceWire® is designed for organizations operating in highly regulated environments, particularly within the life sciences. This includes pharmaceutical, biotechnology and medical device clinical organizations, such as contract research organizations (CROs), contract development and manufacturing organizations (CDMOs) and biologics companies, as well as organizations that must comply with Good Practice (GxP) requirements and global regulatory expectations.
- Can ComplianceWire® integrate with other enterprise systems, such as HRIS or document management tools?
Yes. ComplianceWire® is system-agnostic and built to fit easily into the systems your team already uses. The software is designed to integrate with a wide range of enterprise programs, including human resources information systems (HRIS), document management systems, and other learning or business platforms. This compatibility helps streamline user management, training assignments and document distribution across the organization.
- How does ComplianceWire® help organizations prepare for audits or regulatory inspections?
ComplianceWire® supports audit and inspection readiness by maintaining validated electronic records, secure audit trails, detailed training and qualification histories. The system includes standard reports commonly requested by regulatory authorities and enables organizations to quickly demonstrate employee training status, qualifications and compliance with applicable regulations. Because ComplianceWire® is the same learning management system used by the U.S. Food and Drug Administration (FDA) to train its investigators, organizations can produce reports in a format familiar to regulators, helping to streamline audits and inspections.
- Does ComplianceWire® support role‑based or automated training assignments?
Yes. ComplianceWire® enables automated, role‑based training assignments using configurable learner attributes, such as role, department, location or other organizational criteria. As employee roles or attributes change, training assignments can be updated automatically to help maintain ongoing compliance.
- What scalability options does ComplianceWire® offer for growing organizations?
ComplianceWire® is built to scale with organizations as they grow, supporting complex organizational structures, multiple locations and extended enterprise learning for contractors, partners or suppliers. The platform is designed to accommodate increasing numbers of users, training programs and regulatory requirements without compromising performance or compliance oversight.
- What reporting and analytics features does ComplianceWire® include?
ComplianceWire® provides real‑time dashboards and reporting tools that give administrators and managers visibility into training completion, qualification status and compliance gaps. Reports can be used to monitor trends, identify risks, and support internal and external audits, as well as external regulatory inquiries.
- What type of support does UL Solutions provide during the implementation of ComplianceWire®?
UL Solutions has a highly tenured in-house team with deep expertise developed through hundreds of ComplianceWire® implementations and system validations. Because our team understands the system inside and out, they are able to provide responsive, hands-on support from the beginning of the project to the end. The result for our customers is a streamlined implementation process.
Ready to boost your life sciences training and e-learning capabilities?
Schedule a demonstration to learn how ComplianceWire can help you navigate complex regulatory requirements.