讓您的團隊隨時了解最新動態
醫療器材和藥廠要隨時掌握持續演變的法規非常困難。醫療技術快速發展,因此公司也越來越需要採用符合性系統,以利保障病人安全。為了幫助您和您的員工了解最新的美國食品藥物管理局 (Food and Drug Administration, FDA) 法規、藥品優良臨床試驗準則 (GCP) 培訓、HIPAAA 培訓、ISO 14971 培訓等,我們打造出 ComplianceWire®。
自動化生命科學學習管理
ComplianceWire 學習和資格管理系統是屢獲殊榮、業界領先的培訓學習管理系統 (LMS),屬於 UL Solutions ULTRUS™ 軟體的一部分,可協助生命科學組織管理符合性和資格事宜。ComplianceWire 原生符合 FDA 21 CFR 11 和 EU 附件 11 電子記錄驗證要求。這項經實證的技術,已應用於全球製藥、醫療器材和生物製藥公司內,同時獲得美國、中國、巴西和印度的全球監管機關的採用。生命科學公司使用 ComplianceWire,員工即可享受強大的自動化培訓功能,進一步提升產品的安全性和效能。
FDA 所使用的學習管理技術值得信賴
如同 FDA 透過其中虛擬大學培訓檢查人員和調查人員,UL Solutions 客戶也能享用相同的技術平台和課程。
線上學習圖書館
高品質、引人入勝的知識和內容資產庫,內含 1,000 多個線上學習培訓模組、400 多門生命科學特定課程以及美國 FDA 和 UL Solutions 共同開發的 52 門課程。
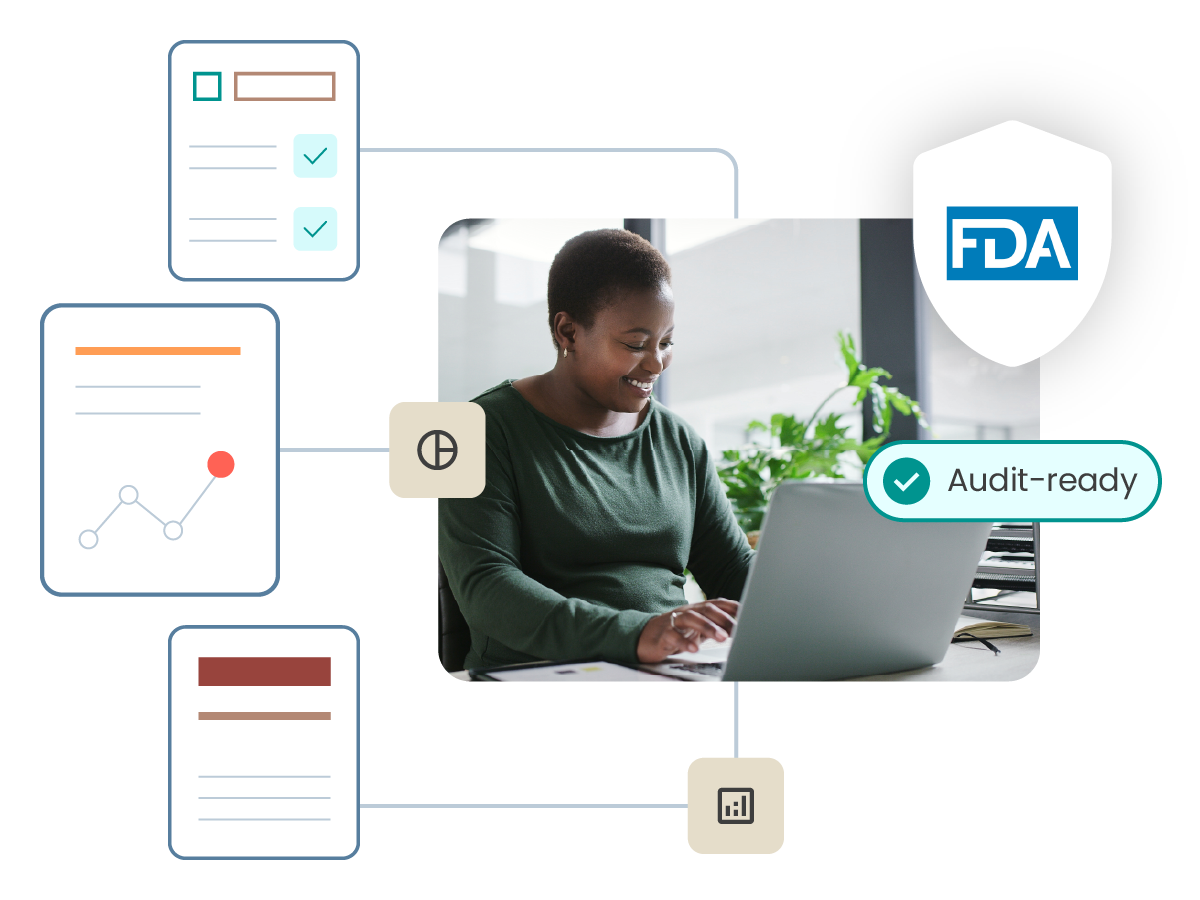
符合 FDA GxP 培訓的要求、21 CFR Part 11、歐盟附錄 11
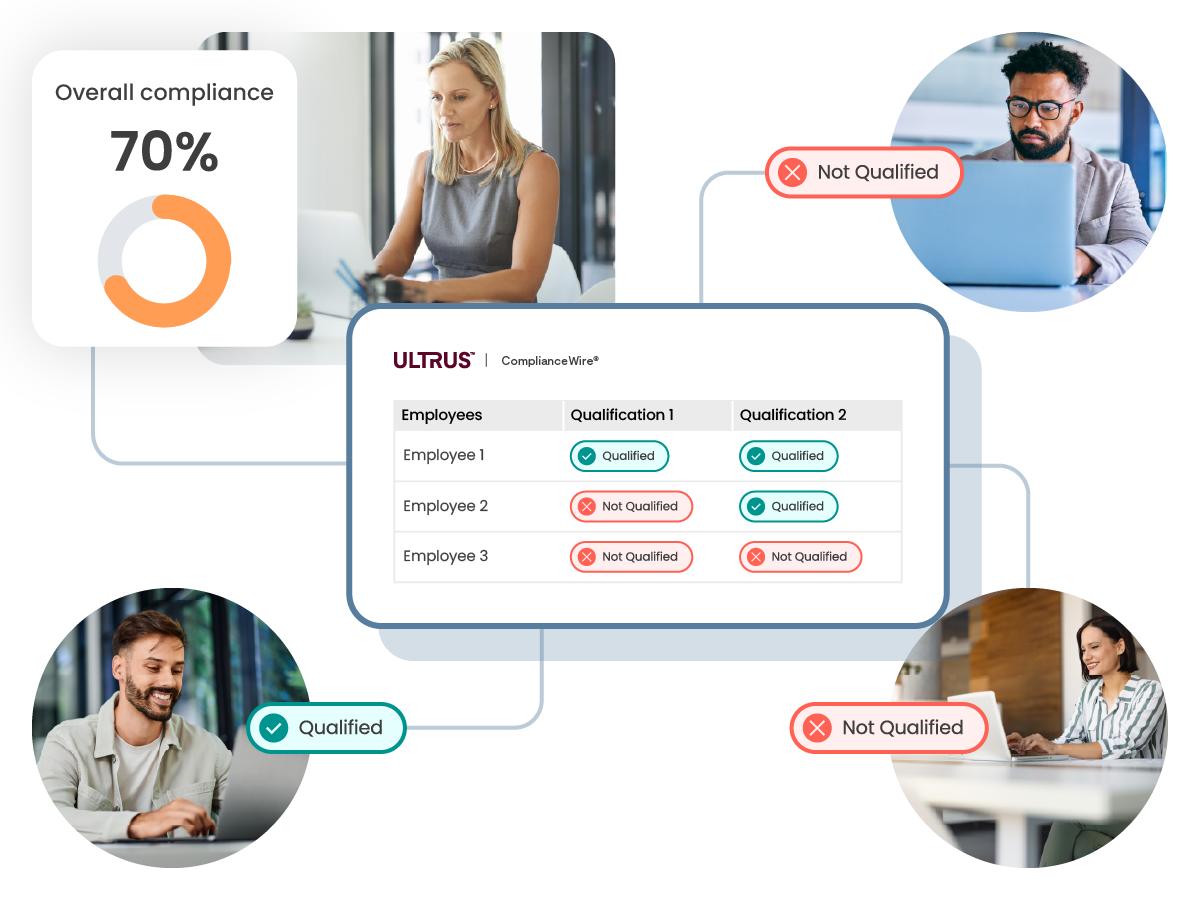
每次實作後,客戶會取得驗證摘要報告、Part 11 白皮書、品質團隊的稽核以及驗證測試文字稿。員工會根據工作職能等標準,自動分配到不同的培訓群組,提升各職責培訓流程的管理效率。自動化版本控制可以降低人為錯誤風險,避免手動執行多個版本協調並更新不斷演變的 SOP 資料庫帶來問題。
ComplianceWire® 和 ULTRUS™ 軟體
ComplianceWire® 現已納入 ULTRUS 軟體,整合了 UL Solutions 的旗艦數位產品,協助客戶應對監管、供應鏈和永續性方面的挑戰。
Frequently Asked Questions
- What types of regulated industries typically use ComplianceWire®?
ComplianceWire® is designed for organizations operating in highly regulated environments, particularly within the life sciences. This includes pharmaceutical, biotechnology and medical device clinical organizations, such as contract research organizations (CROs), contract development and manufacturing organizations (CDMOs) and biologics companies, as well as organizations that must comply with Good Practice (GxP) requirements and global regulatory expectations.
- Can ComplianceWire® integrate with other enterprise systems, such as HRIS or document management tools?
Yes. ComplianceWire® is system-agnostic and built to fit easily into the systems your team already uses. The software is designed to integrate with a wide range of enterprise programs, including human resources information systems (HRIS), document management systems, and other learning or business platforms. This compatibility helps streamline user management, training assignments and document distribution across the organization.
- How does ComplianceWire® help organizations prepare for audits or regulatory inspections?
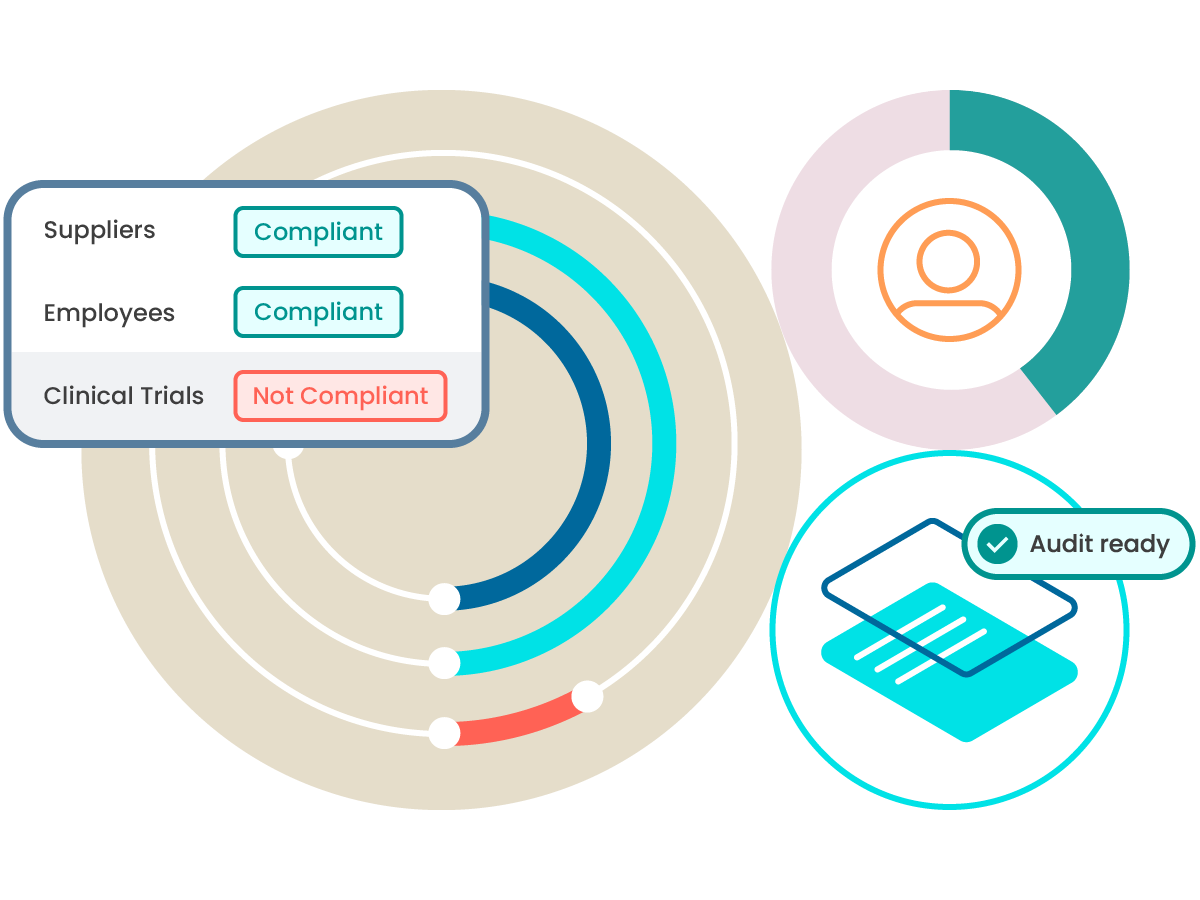
ComplianceWire® supports audit and inspection readiness by maintaining validated electronic records, secure audit trails, detailed training and qualification histories. The system includes standard reports commonly requested by regulatory authorities and enables organizations to quickly demonstrate employee training status, qualifications and compliance with applicable regulations. Because ComplianceWire® is the same learning management system used by the U.S. Food and Drug Administration (FDA) to train its investigators, organizations can produce reports in a format familiar to regulators, helping to streamline audits and inspections.
- Does ComplianceWire® support role‑based or automated training assignments?
Yes. ComplianceWire® enables automated, role‑based training assignments using configurable learner attributes, such as role, department, location or other organizational criteria. As employee roles or attributes change, training assignments can be updated automatically to help maintain ongoing compliance.
- What scalability options does ComplianceWire® offer for growing organizations?
ComplianceWire® is built to scale with organizations as they grow, supporting complex organizational structures, multiple locations and extended enterprise learning for contractors, partners or suppliers. The platform is designed to accommodate increasing numbers of users, training programs and regulatory requirements without compromising performance or compliance oversight.
- What reporting and analytics features does ComplianceWire® include?
ComplianceWire® provides real‑time dashboards and reporting tools that give administrators and managers visibility into training completion, qualification status and compliance gaps. Reports can be used to monitor trends, identify risks, and support internal and external audits, as well as external regulatory inquiries.
- What type of support does UL Solutions provide during the implementation of ComplianceWire®?
UL Solutions has a highly tenured in-house team with deep expertise developed through hundreds of ComplianceWire® implementations and system validations. Because our team understands the system inside and out, they are able to provide responsive, hands-on support from the beginning of the project to the end. The result for our customers is a streamlined implementation process.
您準備好提升生命科學培訓和線上學習能力了嗎?
預約示範,了解 ComplianceWire 如何協助您因應複雜的監管要求。