Create and customize e-learning courses for regulated and non-regulated industries — quickly and professionally
It's increasingly common for product managers, subject matter experts, operations leadership, department administrators and other stakeholders within organizations to be responsible for producing employee learning and process training courseware. But without instructional design or multimedia development expertise, they can find course-building and maintenance to be an overwhelming task and prohibitively expensive to outsource.
Course Create, part of ULTRUS™ software from UL Solutions, empowers your leaders to collaboratively develop courses anytime, anywhere to custom-fit to your organizational needs with our digital sharable content object refernce model (SCORM) authoring tool.
Course Create is simple, but not simplistic. Its breakthrough innovation lies in its ability to blend simplicity with advanced functionality.
With Course Create, you can:

Easily convert PPT slides into e-learning modules
Make new courses or edit courses from UL Solutions’ extensive training library
Set up fully web-responsive and mobile-ready courses

Support versioning
Assign course development roles to streamline workflows
Deliver multimedia content on the ComplianceWire® system worldwide with cloud-based hosting services
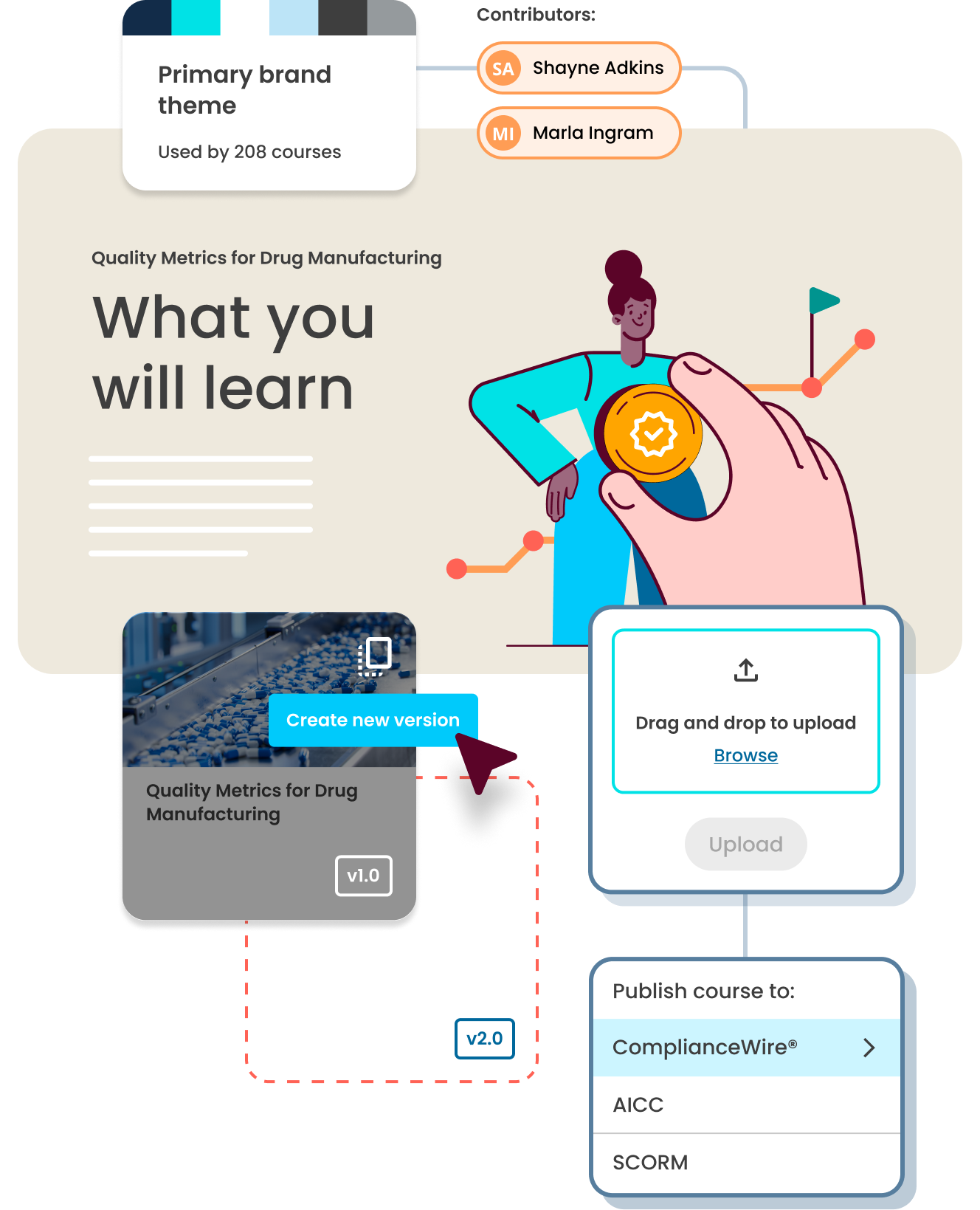
From concept to delivery in five easy steps
Select a design theme and personalize it to meet your branding needs
Add brand-specific logos, fonts and colors to your course. Update your entire training catalog’s branding simply by editing a single course.
Insert your own content or adapt a course from our e-learning library
Add your content slide by slide or choose from the UL Solutions library of up-to-date course titles, created by our Content Solutions department.
Enliven your training with interactive media
Enhance the learning experience by uploading videos, audio, info pop-ups, information trees, quizzes, product logos, third-party hyperlinks and images.
Collaborate easily with cloud-based tools
Work with your content creators worldwide and manage editing, quality and approval processes.
Easily publish to your LMS, or ours
With just one click, publish directly to ComplianceWire® Learning and Qualification Management System or any AICC, SCORM-compliant learning management system (LMS).
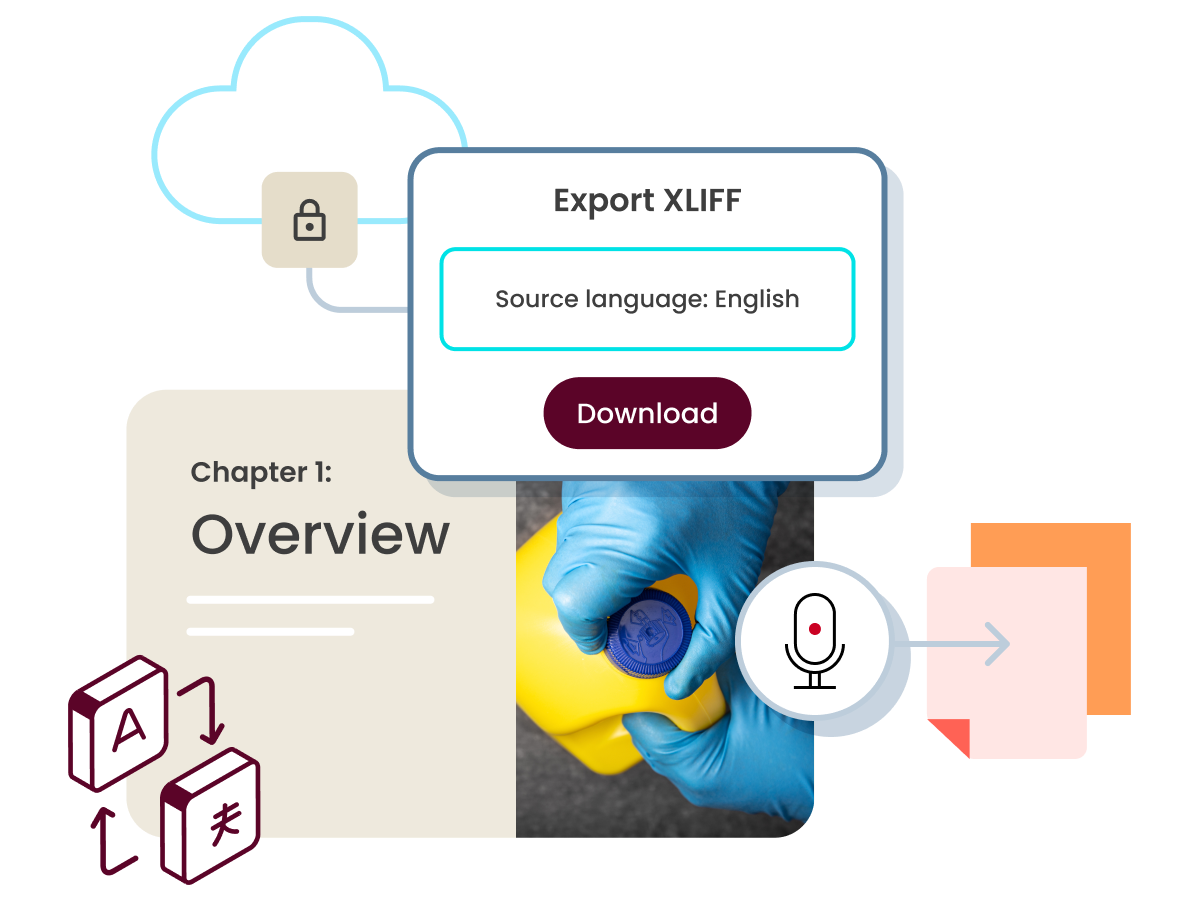
Additional Course Create assets
- Automated course narration implementation via batch uploading of narration files, or record your own narration directly within the tool
- Secure data storage
- Secure cloud-based delivery and storage of your course assets, such as text, images and videos with low latency and high transfer speeds, all within a developer-friendly environment
- Disseminate a course in multiple languages.
- With the help of Localization Interchange File Format (XLIFF), you can create multiple language versions of the same course.
The ease of single-vendor partnership
When it comes to troubleshooting, turning to a single point-of-contact provider makes problem resolution faster and more streamlined, as opposed to coordinating with multiple vendors who are familiar only with their piece of the learning solutions puzzle.
Course Create: your course-building secret weapon
Disseminating accurate, up-to-date training in a constantly changing marketplace is complex, even with today’s sophisticated software offerings.
UL Solutions offers of ready-to-use online training courses that simplify the development of your employee training process, with the responsive design fluidity to match your learners’ devices of choice. Course Create makes the delivery even more powerful by facilitating the creation and customization of engaging content that meets your employees’ learning needs and fulfills compliance requirements.
ComplianceWire triumphs at Brandon Hall Technology Excellence Awards
"The UL Solutions ComplianceWire® division is honored to receive the Gold Award for Best Advance in Content Authoring Technology. With the addition of the ComplianceWire Create authoring suite, we can help the industry generate more effective and engaging learning experiences."
Colleen LoConte, General Manager, ComplianceWire®
Course Create and ULTRUS™ software
Course Create is part of ULTRUS software, which brings together digital offerings from UL Solutions to help customers manage regulatory, supply chain and sustainability challenges.

UL Solutions Create™ DIY e-Learning content creation