December 15, 2020
As vaccines to prevent COVID-19 infections and end the coronavirus pandemic begin to achieve regulatory approvals and commercialization worldwide, pharmaceutical manufacturers, distributors and other stakeholders must now focus their efforts on meeting significant logistical challenges to ensure packaging, transport and delivery of these vaccines without compromising their safety, security or effectiveness.
Coronavirus vaccines require refrigeration, for some products as low as minus 70 degrees Celsius; cold chain management, whereby adequate refrigeration levels are maintained throughout manufacturing, storage and distribution of vaccines, ensures integrity of vaccine compounds via specialized packaging as well as refrigeration and freezer devices. However, ensuring effective cold chain management for COVID-19 vaccines entails many moving parts, as well as particular requirements and constraints around temperature maintenance for transport and storage of pharmaceutical products. Currently, no comprehensive consensus safety, security or performance standard exists that addresses cold chain pharmaceutical transport. Developing such a standard, as well as performance test protocol specifically targeting cold chain processes for vaccines and other pharmaceutical products, will require a convergence of multiple disciplines, including biocompatibility, sterilization, wireless and IoT cybersecurity. In the meantime, cold chain process participants must rely on patchworks of existing safety, security and performance standards, guidelines and best practices.
Below, UL discusses challenges for various stakeholders involved in different aspects of the coronavirus vaccine cold chain process, as well as possible solutions based on existing as well as potential processes, standards and testing.
Pharmaceutical manufacturers: Track and trace
Pharmaceutical companies producing coronavirus vaccines will require adequate visibility to track and trace shipments from their production facilities to final destinations. Companies will also need to obtain temperature readings of vaccine shipments at any point during transport, which will rely on connected sensors and systems. To facilitate these capabilities, manufacturers should consider partnering with qualified third parties to establish proprietary test protocols, understand and mitigate cybersecurity risks for connected systems, as well as determine the appropriate threshold and tolerance criteria necessary for performance testing. Cold chain process protocols and audits will also be necessary to monitor supply chain integrity.
The need for certification for vaccine storage and transport at extremely low temperatures may furthermore necessitate field evaluations and additional labeling requirements; these steps would help pharmaceutical manufacturers demonstrate cold chain assurance to other cold-chain stakeholders as well as healthcare providers and patients.
Additional challenges pharmaceutical companies will need to manage in their cold chain efforts include:
- Integrity maintenance (minimizing risk of failure points)
- Developing processes for identifying vaccines that are about to be exposed to unsuitable temperatures
- Contingency planning and alternate supplier identification for shortages of supplies such as dry ice, refrigeration units, sensors, etc.
- Cybersecurity safeguards and response management to bad actors who might try to steal valuable information or disrupt manufacturing operations.
- Safeguards to prevent unauthorized persons from accessing and using vaccines
- Methods to enable end-users to recognize and dispose of counterfeit vaccines
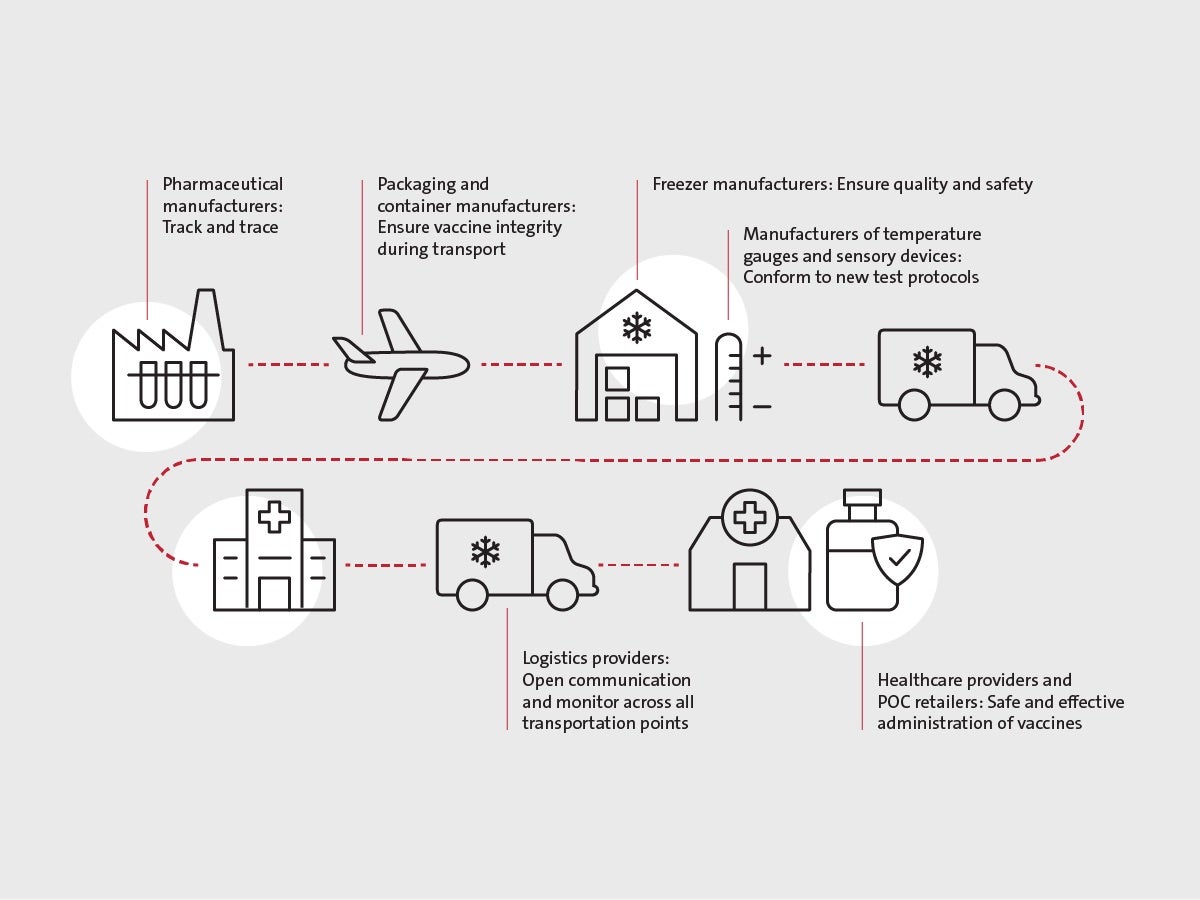
Manufacturers of temperature gauges and sensory devices: Conformance to new test protocols
Effective cold chain processes will also involve laboratory equipment manufacturers producing portable temperature gauges and sensory devices with remote reporting capabilities. These companies may need to demonstrate conformance to new test protocols covering:
- Temperature sensor sensitivity
- Wireless connectivity
- Cybersecurity posture of devices and software solutions
- Immunity to radio frequency noise
- Reliability
- Interoperability
Cybersecurity risk management in particular has already emerged as a key concern in the coronavirus vaccine cold-chain process; various cold-chain participants have already undergone cyberattacks including phishing campaigns, according to IBM. Understanding which cybersecurity guidelines, frameworks or standards are most applicable is of key importance. One example is conformity and testing under cybersecurity standards, such as UL 2900-1, the Standard for Software Cybersecurity for Network-Connectable Products, which can help cold-chain process players identify cyber threats and mitigate associated risks for connected products and systems. Another example of cybersecurity assurance is security assessments of software and hardware suppliers as part of developing these connected products and systems.
Other factors to be addressed by these manufacturers include setting up process evaluations for chains of custody to properly track and document cold-chain materials throughout the storage, transport and distribution of vaccines, as well as for chains of conditions to support consistent vaccine temperatures and related factors during shipments.
Packaging and container manufacturers: Ensuring vaccine integrity during transport
Laboratory equipment manufacturers specializing in vials, climate-controlled packaging and related materials will need to ensure — and demonstrate — that their equipment preserves the efficacy of vaccine doses during transport and storage.
Key questions these manufacturers will need to address include:
- Are cleaning and sterilization of vials part of the initial packaging process?
- How will sterilized vials be filled with sterile vaccines and cooled to required temperatures?
- How will cooled vials be packaged, stored and transported under continuous temperature monitoring?
- How will disposal of assets (vaccine packaging materials, vials, etc.) be managed once vaccines have been distributed?
Existing standards and guidelines can help laboratory equipment manufacturers address some of these requirements. The World Health Organization, for example, has already established cold-chain vaccine packaging and shipping guidelines. In addition, applicable international standards may include IEC 61010-2-011, Safety Requirements for Electrical Equipment for Measurement, Control, and Laboratory Use; ISO 11135-1, ISO 11137-1 and ISO 17665-1 sterilization standards; and ISO 11607-1:2006 for packaging.
Freezer manufacturers: Ensuring quality and safety
For manufacturers of hospital-grade freezers, adhering to safety and performance requirements plays a crucial role in the cold chain process. Quality of freezer cooling properties as well as safety measures for operators as well as buildings must be ensured by these companies; fortunately, several standards and guidelines already exist for testing and certification of such products:
- UL 471, the Standard for Commercial Refrigerators and Freezers
- UL/IEC 60335-1, the Standard for Safety of Household and Similar Appliances, Part 1: General Requirements
- UL/IEC 60335-2-89, the Standard for Particular Requirements for Commercial Refrigerating Appliances with an Incorporated or Remote Refrigerant Unit or Compressor
- IEC 60601-2-011, Safety Requirements for Electrical Equipment for Measurement, Control, and Laboratory Use
When incorporating IoT connectivity into hospital-grade freezers, it is important to develop the right processes to ensure cybersecurity risk is managed across the product lifecycle and across hardware and software components. Cybersecurity assurance across the supply chain is also important to address, with security assessments of software and hardware component vendors, as part of developing these crucial connected products and systems.
Logistics providers: Open communication and monitoring across all transportation points
Logistics companies that will be responsible for warehousing and transporting temperature-sensitive vaccine shipments are tasked with crucial cold-chain process responsibilities, including confirming that communications with all destination points, and that temperature sensor readings occur when and as expected during transportation.
No standards regarding refrigeration transport have yet been established, potentially necessitating development of proprietary test protocols. However, logistics providers may choose to submit their cold chain transportation approaches for process audits under ISO 13485:2016 or ISO 9001:2015 quality management system standards.
Logistics providers may also utilize the standard ASTM D4169-16, Standard Practice for Performance Testing of Shipping Containers and Systems, for simulated testing of transport and distribution of vaccine shipments.
A strong cybersecurity posture is also important for the connected products and systems that communicate data in the cold-chain process. Understanding the key cybersecurity gaps and risks, and developing cybersecurity assurance of the system can both mitigate key business risk as well as help drive confidence with stakeholders.
Finally, logistics providers should consider quality system audits that factor in variable vaccine transportation conditions such as pressurized versus unpressurized air freight cabins, as well as establish safety inspection processes for lithium-ion batteries powering refrigeration units during cold-chain transport. Field evaluation and labeling of refrigeration systems and mobile refrigeration containers may offer immediate cold chain management options.
Healthcare providers and point-of-care retailers: Safe and effective administration of vaccines
Healthcare providers including hospitals, clinics and point-of-care (POC) retail pharmacies on the receiving end of cold chain vaccine shipments will be responsible for loading syringes and administering doses to patients. Providers must ensure their clinicians properly dispense vaccine dosages to patients as well as record any follow-up procedures.
Healthcare providers should utilize appropriate human factors engineering and usability guidelines and best practices, and ensure adherence to clear indications for use (IFU) to meet their responsibilities to patients.
Healthcare providers should also develop internal frameworks, governance and policy for the cybersecurity of their connected products and systems. This also includes security assessments of their software and hardware supply chain, in order to help ensure availability and mitigate key business risks for their crucial vaccine assets.
Ultimately, healthcare providers ad POC retailers face enormous pull-through pressures as these stakeholders will be responsible for dispensing vaccines to patients. POC retailers in particular may rely on their own logistics networks and source their own supplies of refrigerator and freezer equipment for vaccine storage; these companies will require field testing and other methods to verify safety and performance of equipment, as well as third-party reviews and assessments of their cold-chain playbooks to address potential gaps and shortcomings.
Conclusion: Challenges immense but not insurmountable
Although cold-chain stakeholders must grapple with myriad significant challenges now that viable COVID-19 vaccinations are available, many applicable safety, security, performance and related standards as well as regulatory and industry guidelines already exist to address some of these issues. Additional testing, monitoring and vulnerability assessment capabilities will likely also become necessary to ensure safe, secure and effective cold-chain management processes. Resources to support these efforts, such as the Centers for Disease Control and Prevention’s Vaccine Storage and Handling Toolkit that compiles best practices and recommendations for vaccine storage and distribution, are already available.
Get connected with our sales team
Thanks for your interest in our products and services. Let's collect some information so we can connect you with the right person.