UL Solutions for Health and Life Sciences
Contact us today to begin your Health and Life Sciences Journey and elevate your brand’s commitment to safety and innovation.
UL Solutions helps health and life sciences companies with:
Testing and certification
Demonstrate compliance with market requirements and industry standards with our independent testing and regulatory certification services.
Regulatory expertise
Simplify market access and reach users worldwide with our global network of regulatory experts and local expertise.

Software security compliance
Identify software vulnerabilities early in the product life cycle and reduce potential risks related to patient safety and data.
Learning management system
Build a qualified workforce and comply with evolving regulations with our automated, role-based and customized training courses.
Supplier risk management
Our supplier qualification software and audit services can help you increase visibility into supplier data, quality and compliance.
ESG management
Our advisory services and software can help you navigate complex ESG frameworks, overcome reporting challenges, and meet your sustainability targets.
What we offer for health and life sciences innovators
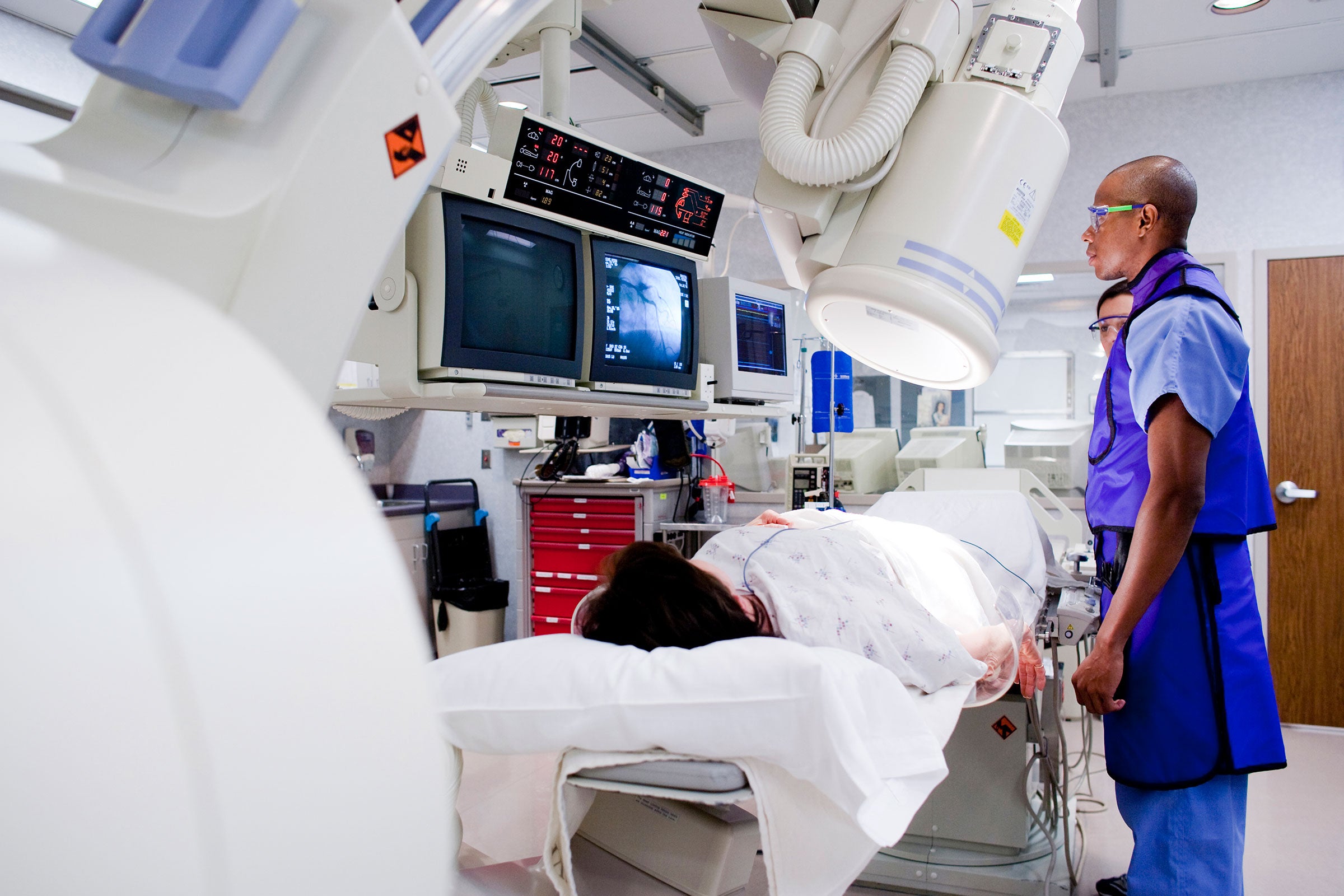
Market access
With our independent testing and regulatory certification services we help health and life sciences companies streamline market access and demonstrate compliance with medical device safety, electromagnetic compatibility, interoperability and biocompatibility standards. With our expertise in human factors engineering and regulatory consulting for medical devices, we help customers deliver usable products.
Cybersecurity
With our expertise in safety science, testing and certification services, and global network of experts, we help customers embed cybersecurity across the product lifecycle of their medical devices and software.
We support enterprises in developing standardized, testable assessment criteria to identify cybersecurity vulnerabilities in their connected medical products and systems, enabling them to manage regulatory and safety requirements.
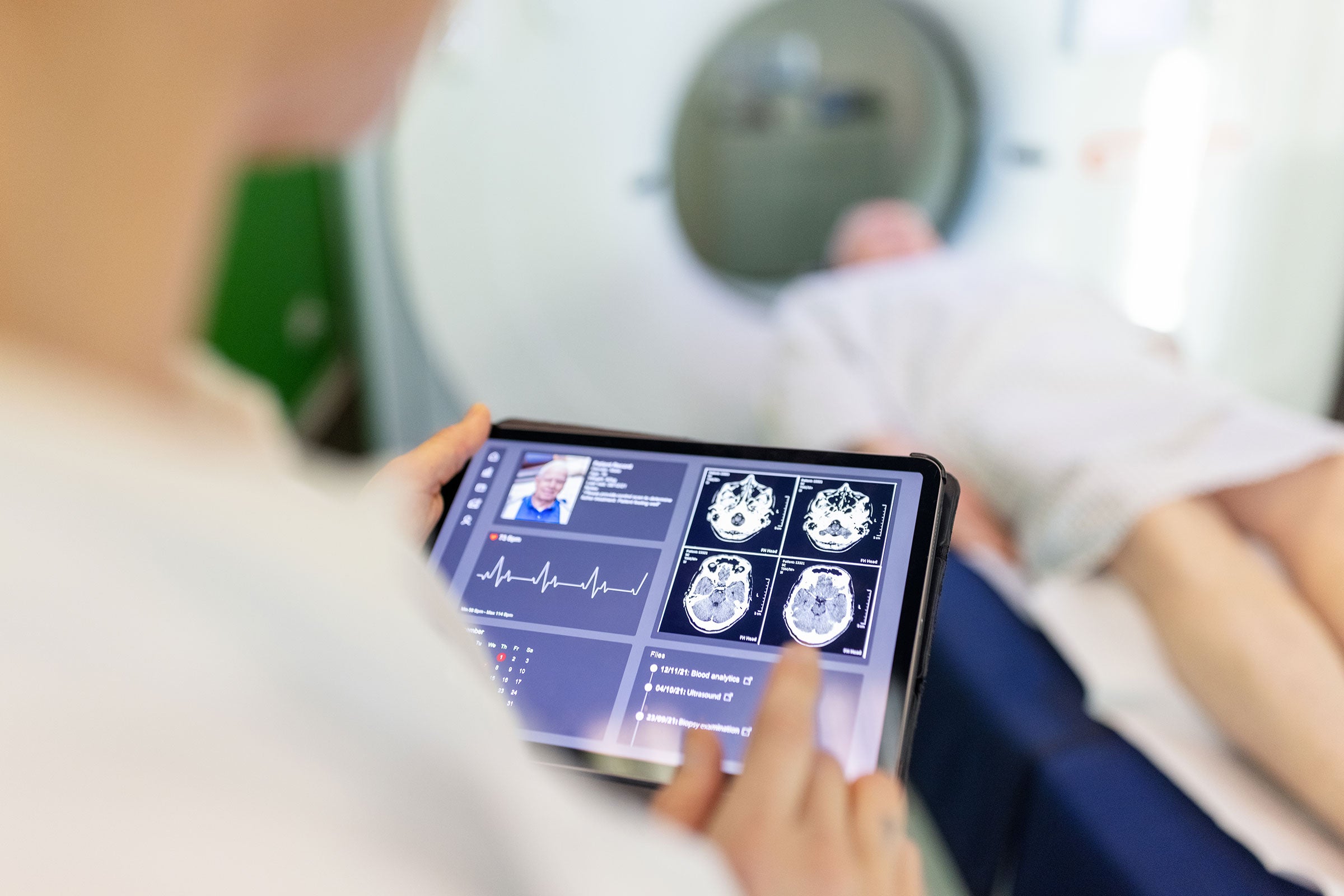

Workforce training and competence
With our automated, role‑based, customized training programs, we help build employee competencies and support compliance with relevant requirements including GxP, HIPAA, OSHA, and ANSI. Our industry‑leading learning management system supports organizations in training their workforces on current quality, compliance, and safety requirements. We also offer on‑demand courses and course‑creation tools to provide organizations with comprehensive training support.
Supply chain resilience
Our supplier qualification software, product insights, and audit services, empower customers with improved visibility into supplier data, quality, and compliance, reducing the risks of product quality issues and supply disruptions.


ESG stewardship
Our ESG management software, advisory and third-party verification readiness services help health and life sciences organizations simplify data collection, reporting, and disclosures, and achieve sustainability goals. In addition, our Zero Waste to Landfill and Landfill waste diversion claims validation and Waste Diversion Certification programs provide trusted, third‑party verification of waste‑reduction performance, helping customers demonstrate meaningful progress toward circularity.
Ready to take the next step?
From demonstrating medical product safety to meeting sustainability goals, UL Solutions provides the safety science expertise and software to reduce non-compliance risks and access global markets with confidence.
* Within UL Solutions, we provide a broad portfolio of offerings to many industries. This includes certification, testing, inspection, assessment, verification and consulting services. In order to protect and prevent any conflict of interest, perception of conflict of interest and protection of both our brand and our customers’ brands, UL Solutions has processes in place to identify and manage any potential conflicts of interest and maintain the impartiality of our conformity assessment services.

