UL Solutions 健康与生命科学
立即联系我们,开启您的健康与生命科学之旅,助力品牌提升安全性与创新性。
UL Solutions 可为健康与生命科学公司提供以下帮助:
测试和认证
借助我们的独立测试与法规认证服务,获取符合市场要求与行业标准的资质证明。
监管专业知识
依托我们的全球法规专家团队与本地专业知识网络,简化市场准入并触达全球用户。

软件安全合规性
在产品生命周期的早期阶段确定软件漏洞,降低患者安全与数据的相关潜在风险。
学习管理系统
通过自动化、角色化的定制培训课程,培养合格员工并符合不断演变的法规要求。
供应商风险管理
借助供应商资质软件与审计服务,强化对供应商数据、质量与合规性的了解程度。
ESG 管理
咨询服务与软件可助力您应对复杂的 ESG(环境、社会和公司治理)框架、克服报告挑战并实现可持续发展目标。
我们为健康与生命科学创新者提供以下服务
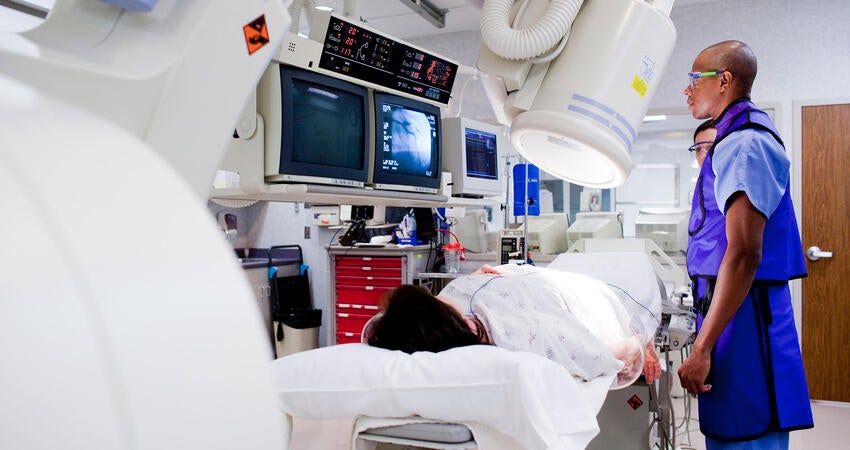
市场准入
助力证明医疗产品的安全性与性能,自动化处理并简化合规事务,弥合人因工程专业知识方面的鸿沟。*
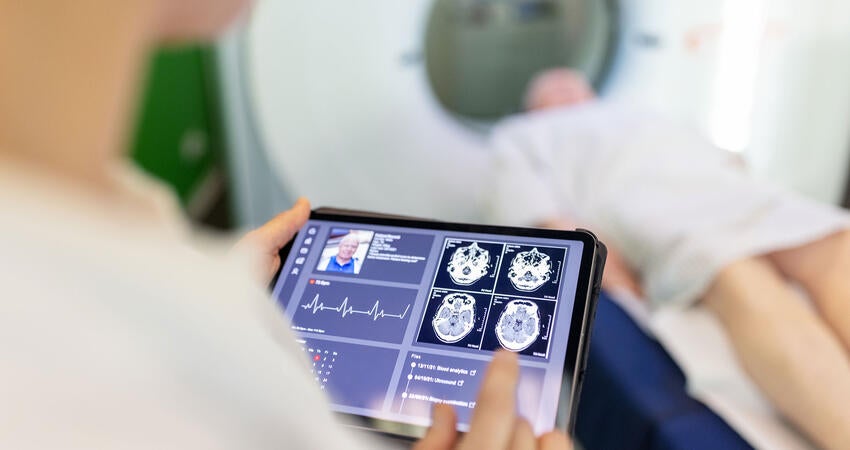
网络安全
依托我们的网络安全专家团队及测试能力,助力证明互联医疗器械设备安全合规。

员工培训与能力培养
借助自动化、角色化的定制培训课程,为员工提供 GxP、HIPPA、OSHA 等合规培训。

供应链韧性
我们的审计服务可验证供应商的资质合规性,助力降低产品质量问题和供应链中断风险。

ESG 监管
利用经碳信息披露项目 (CDP) 认可的 ESG 软件,简化数据收集、报告和披露活动。
What we offer for health and life sciences innovators
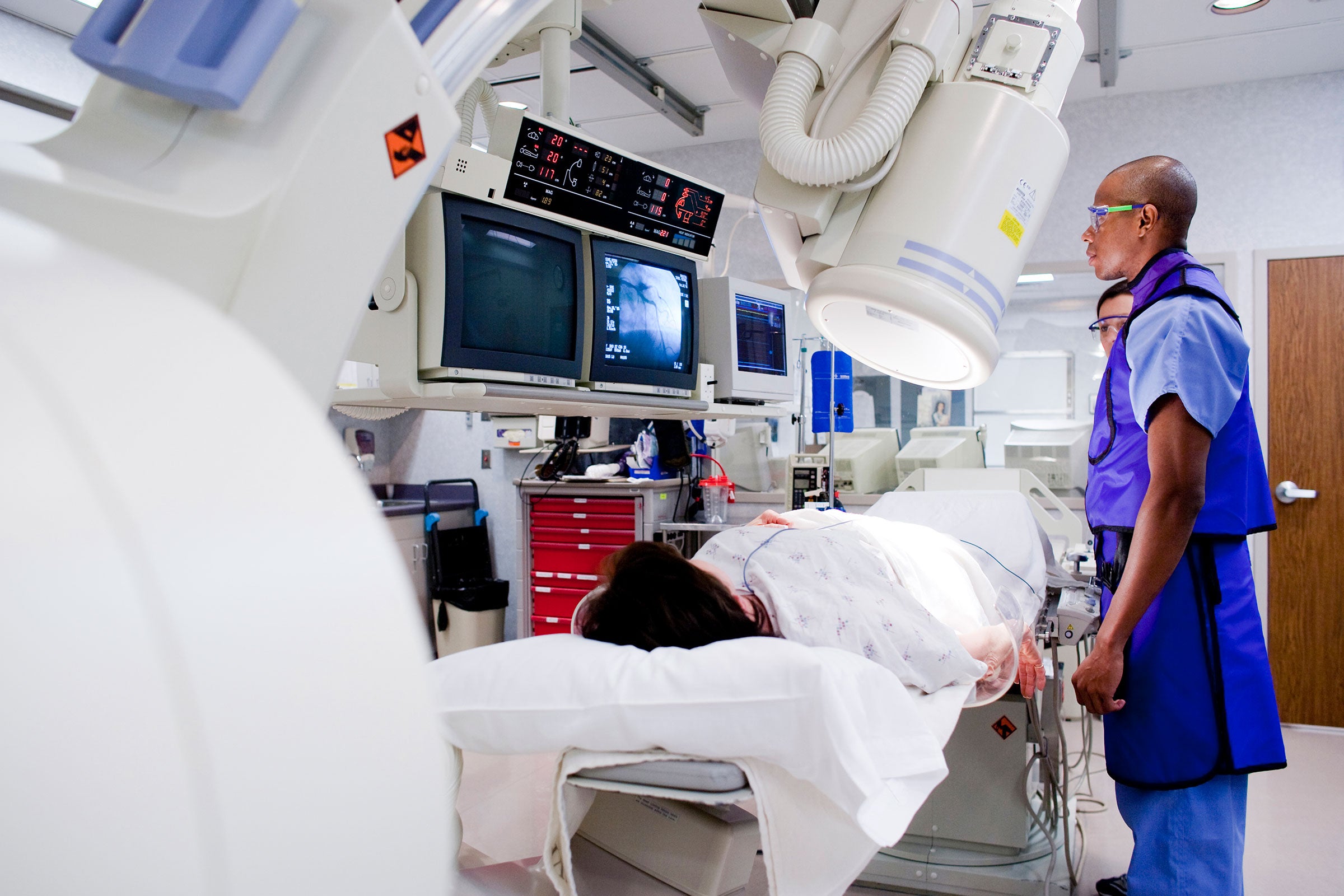
Market access
With our independent testing and regulatory certification services we help health and life sciences companies streamline market access and demonstrate compliance with medical device safety, electromagnetic compatibility, interoperability and biocompatibility standards. With our expertise in human factors engineering and regulatory consulting for medical devices, we help customers deliver usable products.
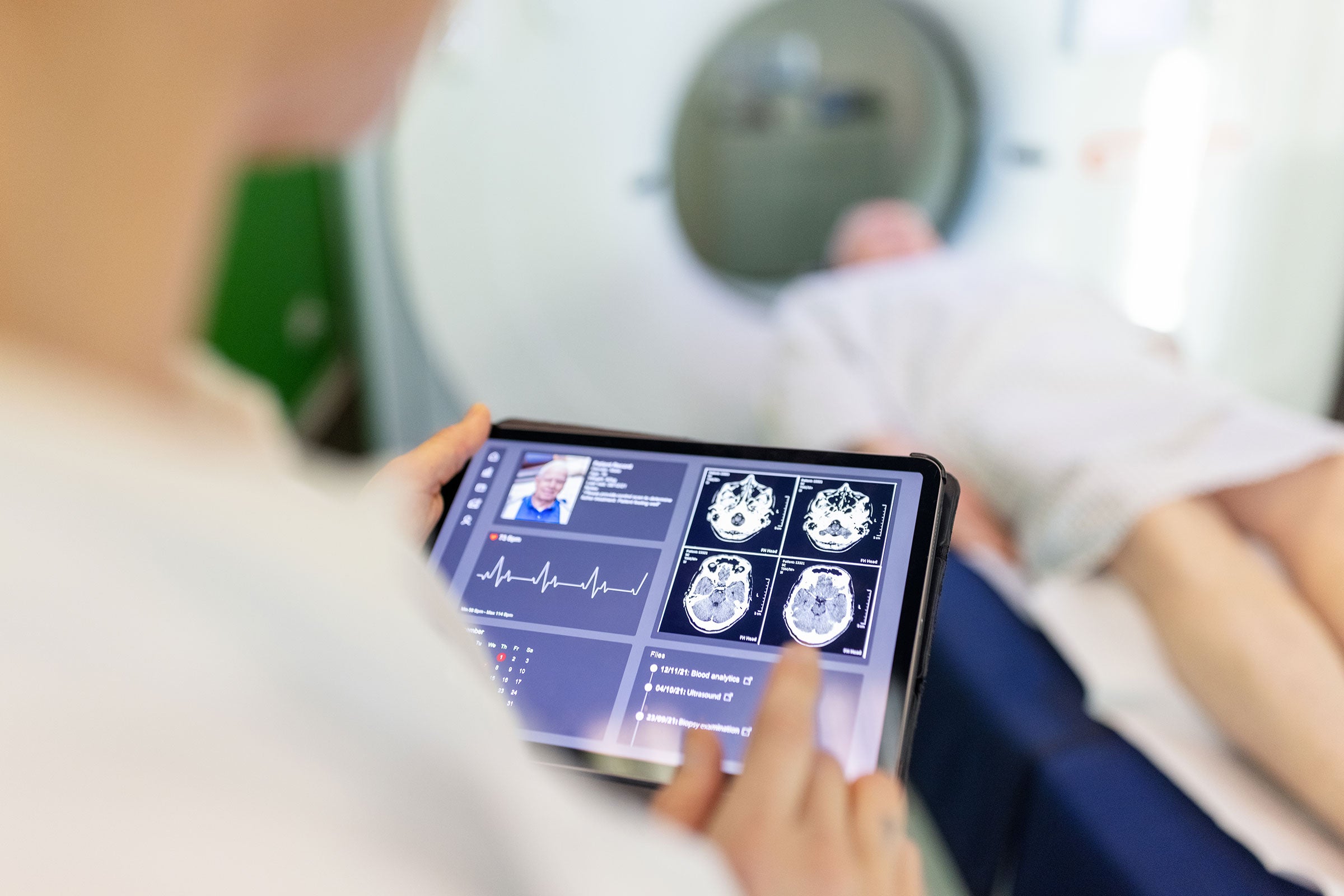
Cybersecurity
With our expertise in safety science, testing and certification services, and global network of experts, we help customers embed cybersecurity across the product lifecycle of their medical devices and software.
We support enterprises in developing standardized, testable assessment criteria to identify cybersecurity vulnerabilities in their connected medical products and systems, enabling them to manage regulatory and safety requirements.

Workforce training and competence
With our automated, role‑based, customized training programs, we help build employee competencies and support compliance with relevant requirements including GxP, HIPAA, OSHA, and ANSI. Our industry‑leading learning management system supports organizations in training their workforces on current quality, compliance, and safety requirements. We also offer on‑demand courses and course‑creation tools to provide organizations with comprehensive training support.
Supply chain resilience
Our supplier qualification software, product insights, and audit services, empower customers with improved visibility into supplier data, quality, and compliance, reducing the risks of product quality issues and supply disruptions.


ESG stewardship
Our ESG management software, advisory and third-party verification readiness services help health and life sciences organizations simplify data collection, reporting, and disclosures, and achieve sustainability goals. In addition, our Zero Waste to Landfill and Landfill waste diversion claims validation and Waste Diversion Certification programs provide trusted, third‑party verification of waste‑reduction performance, helping customers demonstrate meaningful progress toward circularity.
准备好进行下一步了吗?
从客户团队赋能、医疗产品安全认证到助力实现可持续发展目标,UL Solutions 都将提供合适的专业知识与软件工具,降低不合规风险,协助客户自信开拓全球市场。
* UL Solutions 面向多个行业提供多样化的服务,包括认证、测试、检验、评估、验证和咨询服务。为了防止利益冲突(或疑似利益冲突)以及保护我们自身及客户的品牌,UL Solutions 制定了相关流程以识别和管理潜在利益冲突,并保持符合性评估服务的公正性。

