Progressive technology and connectivity represent the present and future of industry product development. Industries have migrated toward new and connected devices to streamline workflows and improve results. The healthcare space is no exception.
Healthcare technology and medical devices are in-step with this product evolution. Medical facilities and caregivers are looking toward these innovations to improve:
- Patient care
- Information access
- Data security
- Product safety
- Result reliability
However, this rapid progress also leaves these devices increasingly susceptible to quality, safety and cybersecurity issues. Manufacturers and developers need to be aware of the inherent risks and current regulations in order to provide compliant and safe products.
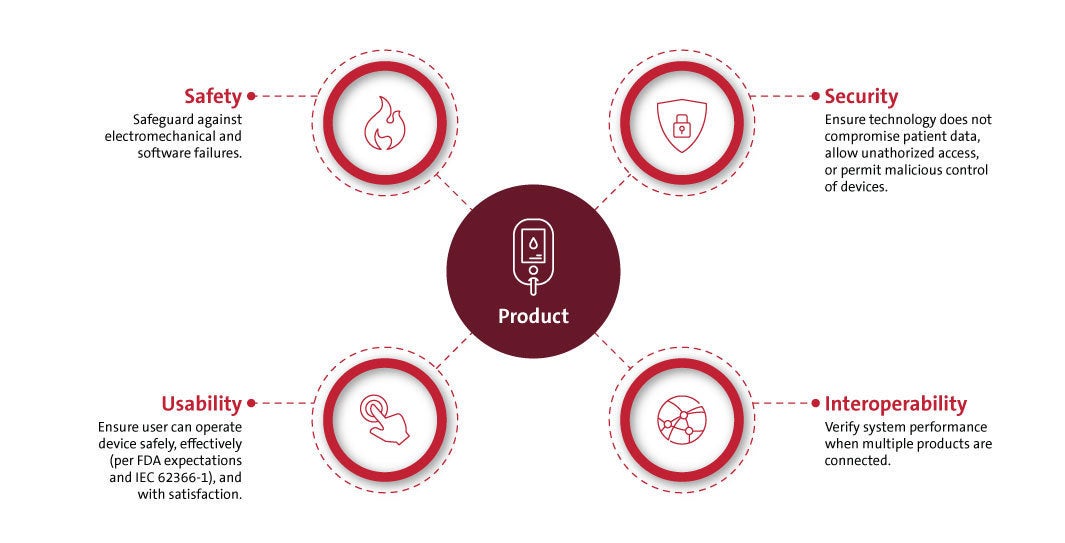
UL Solutions offers safety and compliance testing and training to take on these challenges. Our engineers are current on global regulations and can provide proper product evaluations and safety tests for healthcare products.
Safety testing
Our expert engineers prioritize wellness and safety through our testing services. We offer total product life cycle testing, risk management evaluations, and quality management support for healthcare devices. These evaluations will examine products and facilities for:
- Electrical safety and performance
- Electromagnetic compatibility (EMC)
- Wireless testing, coexistence and Global Market Access Support
- Wired connectivity testing (eg. USB, HDMI, Ethernet)
- Biological evaluation, biocompatibility, microbiological tests, and sterilization services
- Cybersecurity
- Interoperability
In addition, we offer safety management software and a training program (UL’s PURE® Software and Training Solutions) to help operations better manage and understand product safety requirements.
Regulatory compliance
Healthcare device regulatory compliance can be extremely difficult to navigate. We work with global manufacturers, developers and end-users, from design through market release on knowing and adhering to the requirements for the latest standards, to help keep their products compliant.
Our worldwide laboratories provide localized testing services to cover international standards though the U.S., EU, Brazil, Japan, China, Australia and many other regions. These standards include:
- UL 2900 ANSI/CAN/UL Standard for Software Cybersecurity for Network-Connectable Products
- UL 2560 Standard for Emergency Call Systems for Assisted Living and Independent Living Facilities
- UL 1431 Standard for Personal Hygiene and Health Care Appliances
- UL 1069 Standard for Hospital Signaling and Nurse Call Equipment
- IEC 61010 Safety Requirements for Electrical Equipment for Measurement, Control, and Laboratory Use
- IEC/AAMI 60601 Medical Electrical Equipment - General Requirements for Basic Safety and Performance
- ISO 14971 Medical Devices - Application of Risk Management to Medical Devices
- ISO 13485 Medical Devices - Quality Management Systems -- Requirements for Regulatory Purposes
Certification services
UL Solutions' certification services include offerings for product testing and certification, auditing, cybersecurity testing, and usability testing. These services also provide active and inactive medical device manufacturers with the training to support your compliance challenges.
Certification services include:
- UL Mark (US NRTL)
- C-UL Mark for Canada
- IECEE CB Scheme
- INMETRO for Brazil
Medical device testing solutions
Our global reach and local presence allow us to deliver innovative, customized solutions to streamline testing and simplify the certification process.
We can tailor the testing to your own specifications, as well as offer expert advice to help ensure the most efficient use of your time and budget. Leveraging our expertise during the development process can help you avoid costly errors in the design phase and speed time to market.
Earning the UL Mark
The UL Mark is a trusted and respected symbol of safety that can distinguish your medtech devices in the marketplace. Bearing the UL Mark indicates your healthcare product has successfully completed our internationally recognized safety testing and certification. To help certify your products:
- We have accredited consumer and medical product testing laboratories around the world, and our reports and accreditations are recognized and accepted globally for comprehensive market access.
- UL participates in the IECEE CB Scheme, evaluating conformance to related electrical standards.
- We offer testing and certification services, and our engineers work with you throughout the production life cycle.
- We have an FDA-recognized consensus UL 2900, ANSI/CAN/UL Standard for Software Cybersecurity for Network-Connectable Products
- Our testing and compliance engineers work closely with standards committees to stay up to date on all the new amendments and upcoming changes. These committees include:
- Association for the Advancement of Medical Instrumentation (AAMI)
- American National Standards Institute (ANSI)
- International Electrotechnical Commission (IEC)
Watch now: Solutions for the Healthcare and Life Sciences Industry
Dedicated to healthcare industry innovation, we leverage decades of technical, regulatory and clinical expertise to help you manage regulatory challenges and bring safer products to market faster.
Get connected with our sales team
Thanks for your interest in our products and services. Let's collect some information so we can connect you with the right person.